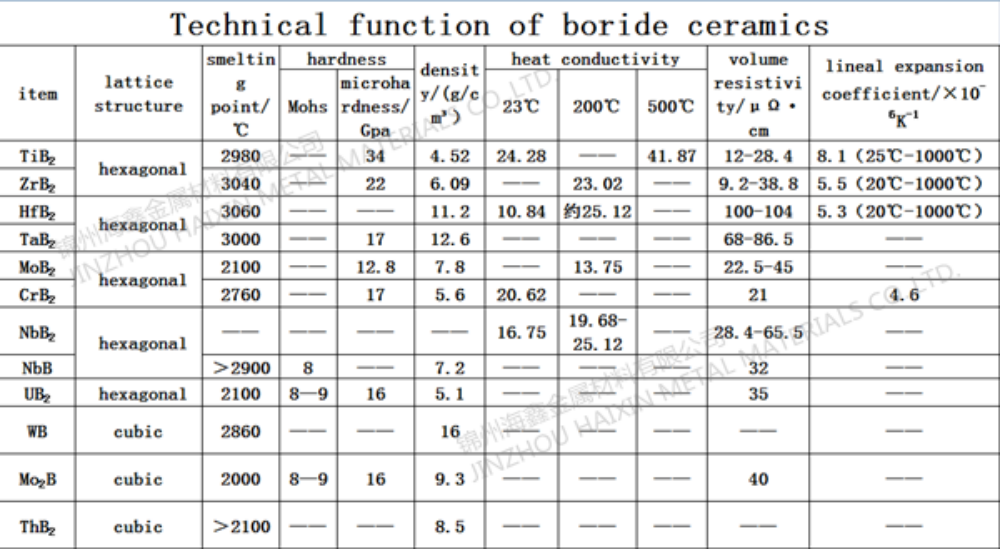
Product Name: Nickel Monoboride (Ni2B)
Specification: 0.8-10um (D50)
Appearance: Irregular
Color: Black Grey
Features: high melting point, high hardness, good chemical stability, high temperature resistance, wear resistance, conductivity, thermal conductivity, and mechanical properties
Application: Catalysts, electronic materials, and other fields

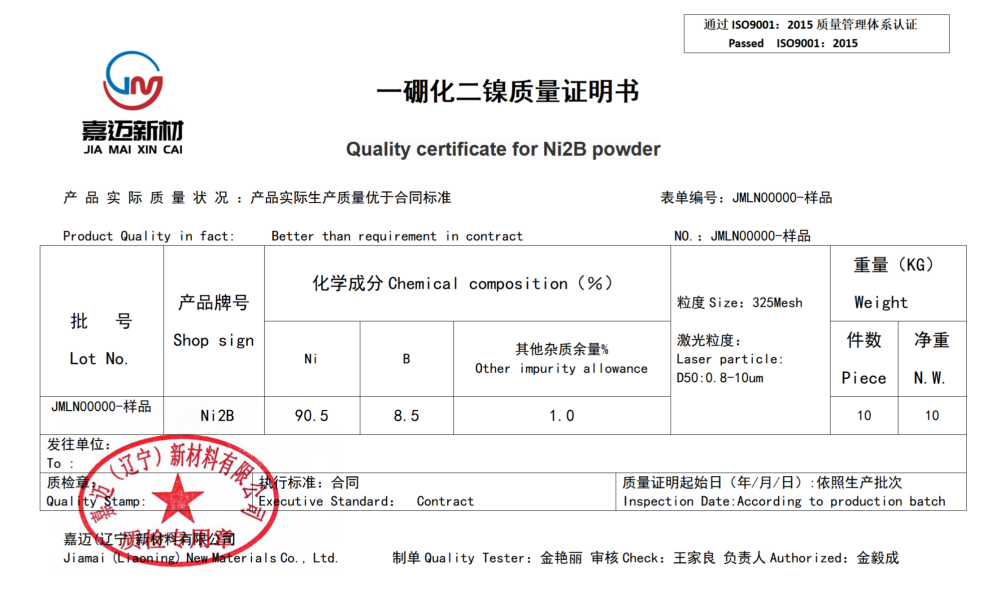

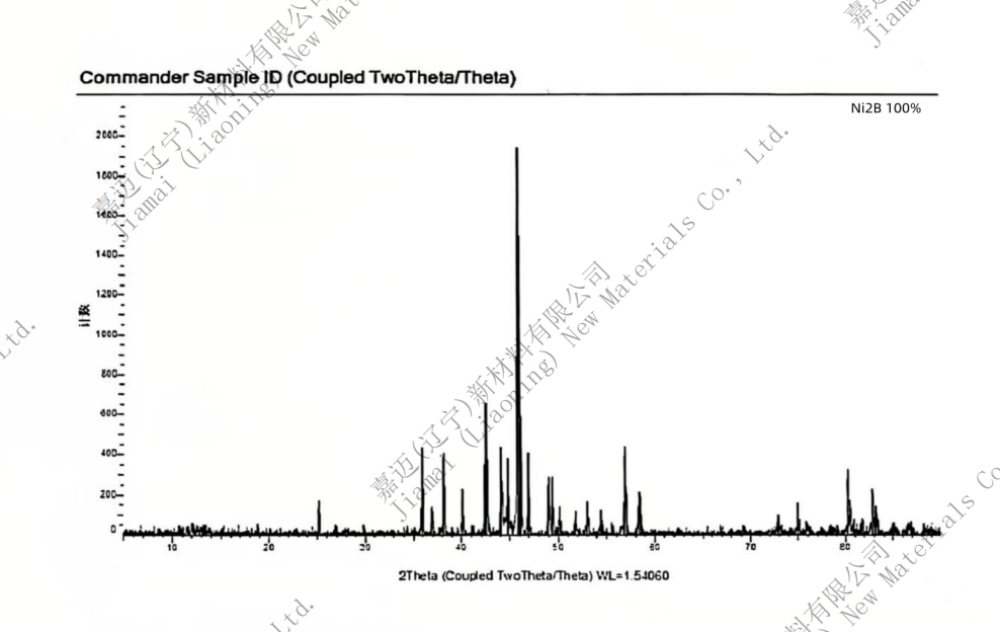



Nickel boride
Chemical formula Ni2B
CAS Number: 12619-90-8
Appearance: Grey black
Density: 7.39g/cm3
Melting point: 1020 ℃
Molecular weight 69.52, solid relative density 7.3918
Has strong magnetism. Soluble in aqua regia and nitric acid. Although stable in dry air, it quickly reacts in humid air, especially in the presence of CO2. Can react with chlorine gas when hot. When heated together with water vapor, nickel oxide and boric acid can be generated.
Application: Nickel boride was originally used as a catalyst for various reactions in a hydrogen atmosphere, and has now been used as an reactant and catalyst in many reactions. The advantages of nickel boride mainly include high hardness, good catalytic effect, high chemical and thermal stability. It has good selectivity and reaction activity in liquid-phase reactions and can be used as a non precious metal hydrogen electrode catalyst and fuel cell trap electrode catalyst.